Fda Class 2 Medical Device Approval Process
The study will examine the program also called the 510k process for medical devices which can be accessed. FDA Commission On Approval Process The US.

Software As Medical Device Samd Classification And Definitions
But to get FDA approval youll need to prove compliance.
Fda class 2 medical device approval process. Found substantially equivalent through the 510 k process. Author Gail A Van Norman 1 Affiliation. Medical devices undergo frequent modifications to their design and materials due to many things.
Step 9 Upon TFDA approval you will be issued a Medical Device Product License valid for five years. And What Is FDA Compliance. FDA approval means that the FDA has formally approved your product in this case your medical device.
Some products ie low. FDA medical device approval process step-by-step guide. Compared to a US 510 k application MDL applications are simpler for Class II devices and about the same for Class III devices.
A primary safeguard in the way FDA regulates medical devices is the requirement that manufacturers must submit to FDA a Premarket Approval PMA application if they wish to market any new products. An Overview of Approval Processes. The De Novo process provides a pathway to classify novel medical devices for which general controls alone or general and special controls provide reasonable assurance of.
Arsip April 2021 49. Label 2018 2019 2020 abortion abroad academy accepts account acid address admission advice africa after agency aggravate aires alamal alamitos alliance allowance aloe alumni. And 3 the humanitarian device exemption HDE process.
There are 3 basic processes to obtain FDA marketing approval for medical devices depending on the nature of the device and the circumstances under which approval is sought. Confirm That You Have a Medical Device Thats Regulated by FDA and Needs a 510k This may seem obvious but the very first step is to confirm that your product is a regulated medical device and needs to go through the 510k approval process. Pay special attention to the three-letter Product Code and seven-digit.
Recalls occur when a medical device is defective when it could be a risk to health or when it is both defective and a risk to. Approximately 4000 510 k applications are submitted to the FDA. 2 Total Product Life Cycle TPLC TPLC Product Code Report.
Renewal documents must be submitted six months before expiration. For Class I devices apply for a Medical Device Establishment License MDEL For Class II III and IV devices apply for a Canadian Medical Device License MDL application for your device. By the numbers the vast majority of medical devices are cleared through 510 k versus approved through the PMA pathway.
The FDA does approve Class III medical devices via the PMA process. Drugs Devices and the FDA. Food and Drug Administration FDA just announced it commissioned the Institute of Medicine IOM to study the pre-market notification program used to review and clear certain medical devices marketed in the United States.
Clinical trial information will be required unless the device is Class II and qualifies for the Simplified Application Route noted above. FDA Approval of Medical Devices JACC Basic Transl Sci. Step 1 Determine the classification of your medical device or in vitro diagnostic IVD device by searching the FDA classification database using relevant search terms or by identifying another predicate device with the same intended use and technology.
Cari Blog Ini Diberdayakan oleh Blogger Gambar tema oleh Igniel. Decide the classification of your device by examining the FDA classification database using relevant search terms or by distinguishing another device with the equivalent planned use and innovation. Division of Anesthesia Respiratory and Sleep Devices DHT1C Division of Anesthesia Respiratory and Sleep Devices DHT1C Submission Type.
Recalls A recall is an action taken to address a problem with a medical device that violates FDA law. For Class III devices a premarket approval application PMA will be required unless your device is a preamendments device on the market prior to the passage of the medical device amendments in. 2 the PMN process.
In most cases the medical device company will consult with the FDA at the beginning of the process in order to choose the correct pathway to prevent lost time and costs. Changes in the supply chain continuous process improvement or to keep pace with. This is important to establish that your device is safe and effective.
Premarket approval PMA is the FDA process of scientific and regulatory review to evaluate the safety and effectiveness of Class III medical devices. 1 the PMA process. Grant special attention to the three-letter Product Code and seven-digit.
The US FDA medical device IVD approval process explained. Class I and a small amount of Class II devices may apply for exemption with the FDA. 3 Basic Pathways to Medical Device Approval.
Applicants must compare their device to one or more similar legally marketed devices and make and support their substantially equivalent.

Fda Medical Device Approval Process Step By Step Guide
Understanding When To Revalidate Your Medical Device Process
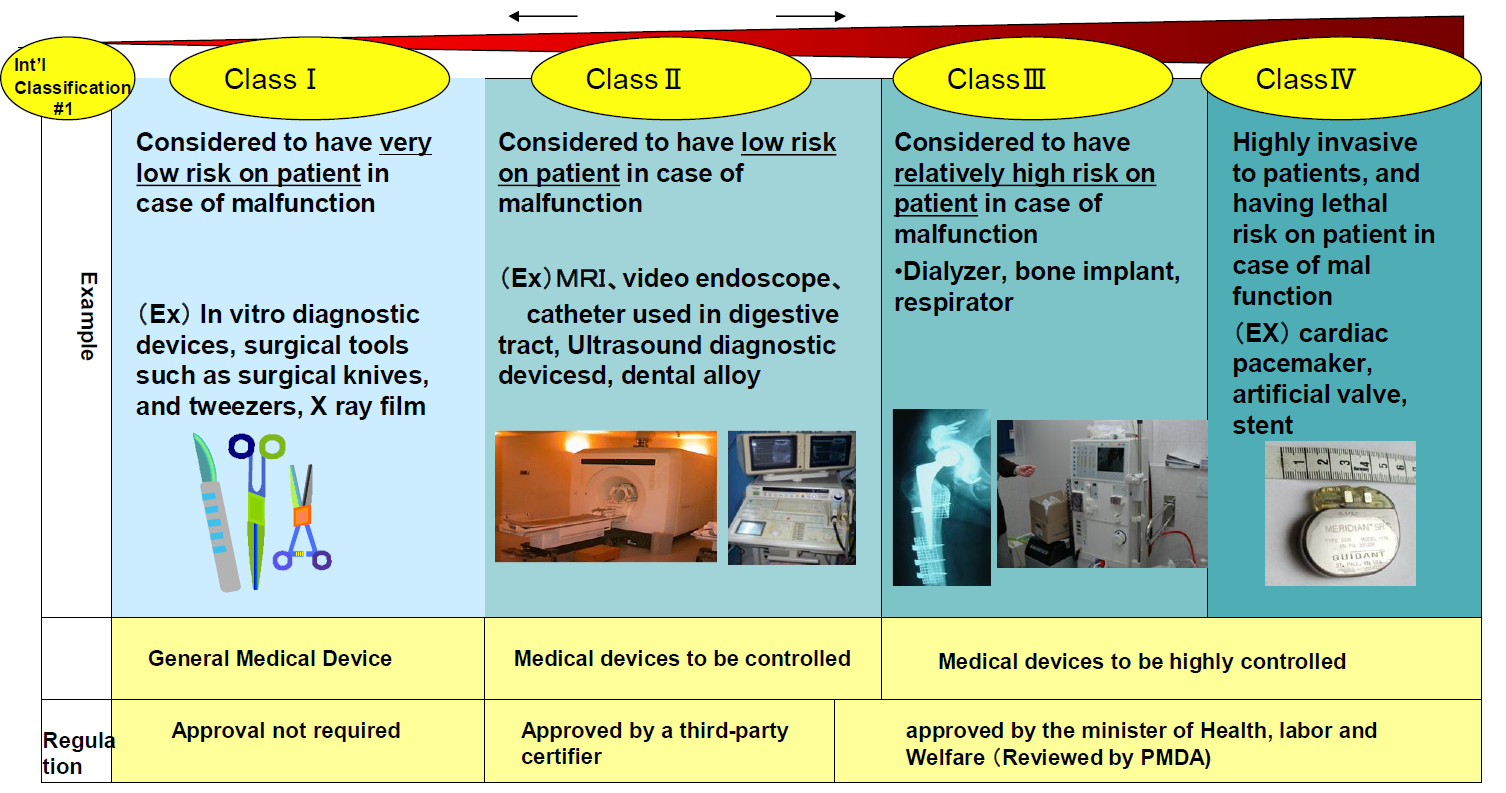
Interoperability Standards For Medical Device Integration In The Or And Issues Relating To International Approval Procedures Part 4 Iscasblog

8 Things To Know About Ce Approval Process For Medical Devices Medicaldevice Cemark Approval Process Regulato Medical Medication Management Medical Device

Medical Device Manufacturing Consultants In India Operon Strategist Medical Device Medical Understanding
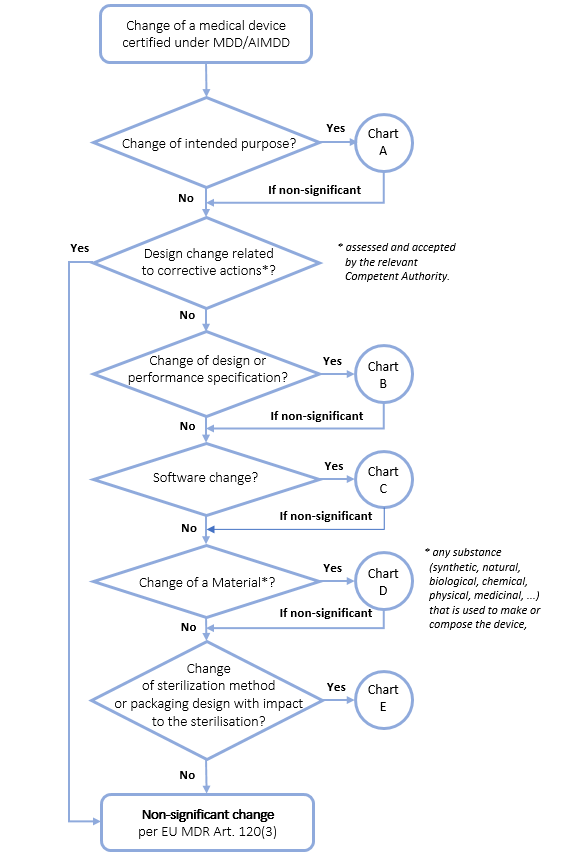
Mdr Guidance On Significant Changes For Medical Devices

Fda Medical Device Approval Process Step By Step Guide
Mexico S Regulatory Process For Medical Devices Mdrc
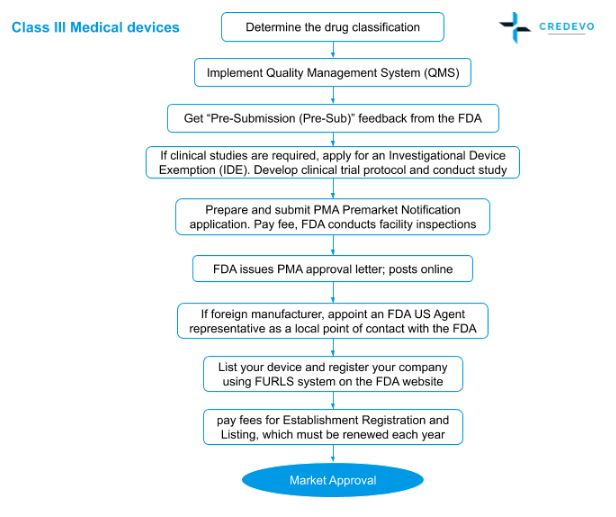
Medical Device Market Approval Process In The United States Credevo Articles
Medical Device Process Validation What You Need To Know
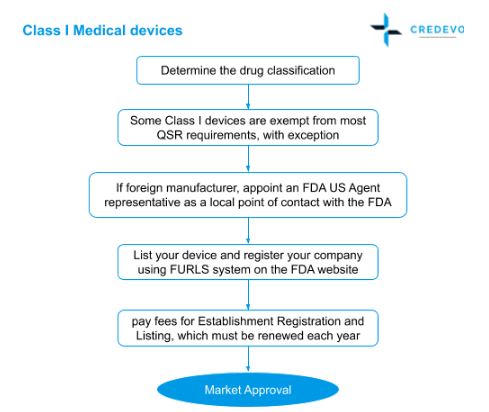
Medical Device Market Approval Process In The United States Credevo Articles

Medical Device Market Approval Process In The United States Credevo Articles

Market Access For Medical Software In The United States Vde Medical Devices And Software

Registration Of Medical Devices In Brazil
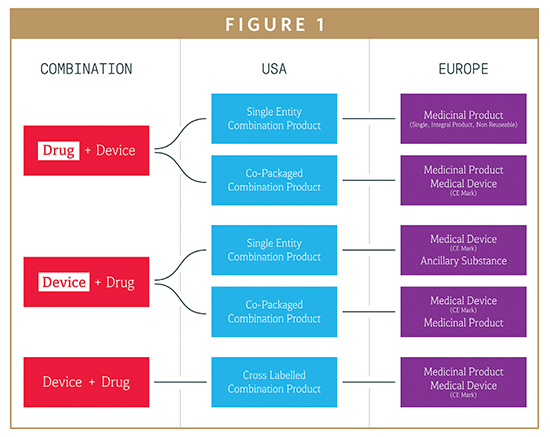
Device Regulations The New Medical Device Regulation The Applicability Of Article 117 To Medicinal Products
Medical Device Process Validation What You Need To Know
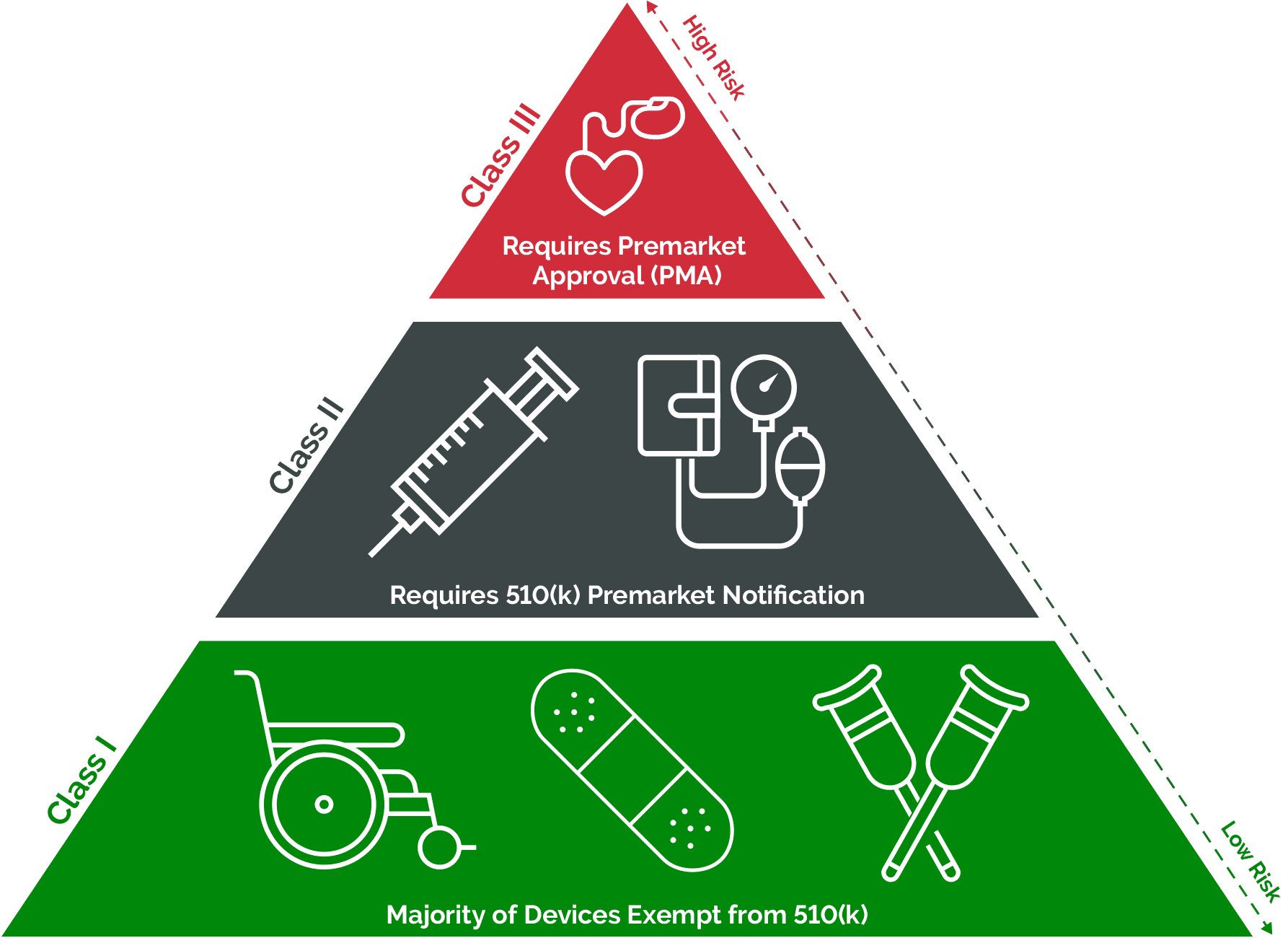
How To Classify Your Medical Device For Fda Approval Arena
5 Top Annual Plan Medical Device Design And Development Process Improvements Starfish Medical
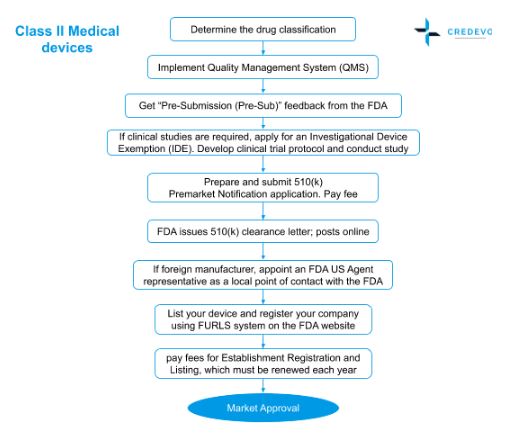
Medical Device Market Approval Process In The United States Credevo Articles
Post a Comment for "Fda Class 2 Medical Device Approval Process"